Juvenile Nasopharyngeal Angiofibroma
Synonyms: angiofibroma, juvenile angiofibroma, juvenile nasopharyngeal angiofibroma, (JNA)
Definition: Juvenile nasopharyngeal angiofibroma (JNA) is a histologically benign , but locally invasive neoplasm occuring almost exclusively in adolescent males. These tumors are highly aggressive and are associated with significant morbidity and mortality due to its tendency to bleed.
History: Juvenile nasopharyngeal angiofibroma has been documented since the time of Hippocrates (4 BC). Infact Hippocrates goes on to describe a polyp in the nose which weeped blood. In 1906 Chareau revived the interest in the study of juvenile nasopharyngeal angiofibroma. Shaheen in 1930 reported the first female patient with juvenile nasopharyngeal angiofibroma. Hondousa recorded the youngest JNA patient (8 years). Figi and Davis (1950) emphasised the role of surgery in the management of JNA. They also suggested that subperiosteal dissection of the mass reduced bleeding to a great extent.
Histopathological studies of JNA tissue was extensively done by Harma (1959).
Anatomy of Nasopharynx: Since JNA involves the nasopharynx, a complete understanding of nasopharyngeal anatomy is a must for the safe management of the problem. This area has been considered as an antomical blind spot (a virtual no man's land) because of its relative inaccessibility. It is very difficult to visualise completely the entire nasopharynx. Cawthorne (1953) remarked "I am never sure whether I will be able to get a really satisfactory view of the whole nasopharynx". The advent of endoscopes have made the task of examination of nasopharynx simpler these days. The recent imaging modalities like CT scan and MRI has further simplified non invasive ways of examining the nasopharynx.
Location: Nasopharynx is located at the confluence of nasal, aural and pharyngeal air passages. It is an unyielding irregular cuboidal box containing
4 x 3 x 2 cm air space ar the base of middle cranial fossa.
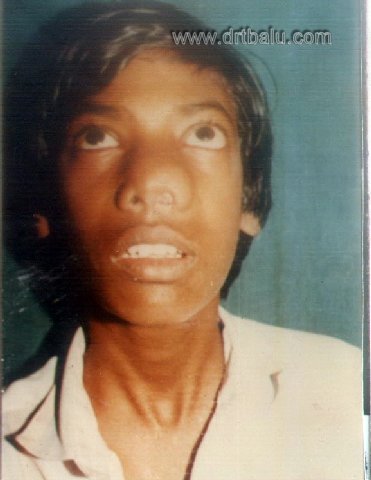
Clinical photograph of a patient with JNA
Diagram showing the Nasopharynx
Nasopharynx is located immediatly behind the nasal cavities and below the body of the sphenoid and basi occiput above the level of soft palate as illustrated in the figure. Posteriorly it is limited by the first two cervical vertebrae. The posterior wall of nasopharynx is about 8 cm from the pyriform aperture along the floor of the nose.
Boundaries of Nasopharynx:
Anterior: Choanal orifice and posterior margin of nasal septum.
Floor: is formed by the upper surface of soft palate in its anterior 2/3 while the posterior 1/3 is formed by nasopharyngeal isthumus.
Roof and posterior wall: is formed by the continuous sloping surface of the body of the sphenoid, the basiocciput and the first two cervical vertebrae up to the level of the soft palate. The upper portion of the posterior wall lies in front of the anterior arch of atlas with a mass of lymphoid tissue embedded in the mucous membrane (adenoid). The prevertebral fascia and muscles separate the adenoid from the vertebrae.
Lateral wall: is dominated by the pharyngeal end of eustachean tube. It is located in the middle of the lateral wall, it is about 1.5 cm equidistant from the roof, posterior wall, choana and the floor. The tubal elevation created by the elastic cartilage of the tube is particularly prominent in its upper and posterior lip. Behind the posterior margin of torus, between it and the posterior wall lies the lateral pharyngeal recess or the fossa of Rosenmuller. Aggregates of lymphoid tissue (Gerlachs tonsil) of variable sizes surround the tubal orifice. This is alsl known as the tubal tonsil.
Fossa of Rosenmuller is situated at a corner between the lateral and dorsal walls. This recess is not obvious in infants, and can measure up to 1.5 cm in depth in adults. It opens into the nasopharynx at a point below the foramen lacerum related to the internal carotid artery.
Boundaries of Fossa of Rosenmuller:
Anterior: Eustachean tube and levator veli palati muscle.
Posterior: Pharyngeal wall mucosa overlying the pharyngobasilar fascia and retro pharyngeal space, containing the retropharyngeal lymph nodes of Rouviere.
Medial: Nasopharyngeal cavity.
Superior: Foramen lacerum and floor of the carotid canal.
Postero lateral (apex): Carotid canal opening and petrous apex posteriorly, foramen ovale and spinosum laterally.
Lateral: Tensor palati muscle, mandibular nerve and the prestyloid compartment of the para pharyngeal space.
Endoscopic view of Nasopharynx
Epithelial lining of nasopharynx: The mucosal lining of nasopharynx is thrown into numerous folds and crypts. The surface area of mucosal llining of nasopharynx is about 50 cm 2 in adults. The mucosa abutting the choanae and immediate nasopharyngeal roof is completely lined by ciliated columnar epithelium. The nasopharyngeal mucosa differs from the rest of the upper respiratory tract in that the subepithelial connective tissue is rich in lymphoid tissue.
To understand the clinical features of mass lesions involving the nasopharynx, a clear understanding of the roof of the fossa of Rosenmuller is important. As already mentioned the roof of fossa of Rosenmuller is formed by the Foramen Lacerum. Through this foramen lacerum passes:
a. Internal carotid artery
b. Greater superficial petrosal nerve
c. The ascending palatine artery sometimes passes through this foramen.
It is through this foramen tumors from fossa of Rosenmuller invades intracranial structures. The 3rd, 4th, 5th and 6th cranial nerves are in juxtaposition of the foramen. These nerves are commonly involved when tumors invade intracranial structures via the foramen lacerum.
Blood supply of nasopharynx:
a. Ascending pharyngeal artery
b. Ascending palatine branch of facial artery
c. Branches of internal maxillay artery
Veins form a plexus which communicate above with the pterygoid plexus and drains into the common facial and internal jugular veins.
Lymphatics: Nasopharynx is richly endowed with lymphatics. They mainly originate in the pharyngeal tonsil and runs laterally and downwards on the pharyngeal aponeurosis, some of them terminating in the median and lateral veins. The collecting trunks terminate for the most part in the upper nodes of the spinal accessory chain, which lie under the upper end of sternomastoid muscle, and also into the jugulodigastric node of the internal jugular chain. From these nodes efferent branches run down to the middle and lower group of nodes of the internal jugular and spinal accessory chains.
Nerve supply: is derived mainly from the pharyngeal plexus which is formed by branches of the 9th and 10th cranial nerves together with sympathetic fibers.
Aetiopathogenesis of JNA:
This relatively rare tumor occurs in the second decade. Almost exclusively adolescent males are affected. The reported rate of incidence varies from 1/6000 (Harma 1959) to 1/50,000 (Hondousa etal 1954). The exact nature of the tumor and its etiology is not well known. Various theories have been propounded to explain the etiopathogenesis of JNA.
Theories of etiopathogenesis of JNA:
Ringertz theory: This theory was proposed by Ringertz in 1938. He believed that JNA always arose from the periosteum of the skull base.
Som & Neffson (1940): believed that inequalities in the growth of bones forming the skull base resulted in hypertrophy of the underlying periosteum in response to hormonal influence.
Bensch & Ewing (1941): thought that the tumor probably arose from embryoninc fibro cartilage between the basi occiput and basi sphenoid.
Brunner (1942): Suggested an origin from conjoined pharyngobasilar and buccopharyngeal fascia.
Marten et al (1948): Proposed a hormonal theory suggesting that these tumors resulted from deficiency of androgens or over activity of estrogens and that the hormonal stimulation is responsible for angiomatous components seen in JNA tissue.
Sternberg (1954): Proposed that JNA could be a type of haemangioma like a cutaneous haemangioma seen in children which regresses with age.
Osborn (1959): Considered two alternatives to explain the etiology of JNA. They proposed that the swelling could be due to either a hamartoma or residual fetal erectile tissue which were subject to hormonal influences.
Girgis & Fahmy (1973): Observed cell nests of undifferentiated epitheloid cells or "Zell ballen" at the growing edge of angiofibromas. This appearance was more or less similar to that of paraganglioma. They considered JNA to be a paraganglionoma.
The most accepted theory is that JNAs originate from sex steroid–stimulated hamartomatous tissue located in the turbinate cartilage. The proposed hormonal influence may explain why (rarely) some JNAs involute after puberty.
Pathophysiology: The proposed origin of the JNA is located along the posterior-lateral wall in the roof of the nasopharynx, usually in the region of the superior margin of the sphenopalatine foramen and the posterior aspect of the middle turbinate. Fetal histology confirms large areas of endothelial tissue in this region. Rather than invading surrounding tissue, this tumor displaces and distorts, relying on pressure necrosis to destroy and push through its bony confines. Intracranial extension is noted in 10-20% of cases.
JNA are seldom seen in children below the age of 8. The rate of growth of tumor and period of maximum development coincides with rate of erectile tissue of penis, both increasing in size during the period of sexual development.
Pathology:
Macroscopic: Grossly, angiofibromas appear as firm slightly spongy lobulated swelling with presence of nodules. The nodularity increases with age. Their color varies from pink to white. The part which is seen in the nasopharynx and which is covered by mucous membrane is invariably pink, where as those parts which have escaped to adjacent extra pharyngeal areas are often white or grey.
On section the tumor is reticulated, whorled or spongy in appearence lacking a true capsule. The edges of the tumor are however, sharply demarcated and easily distinguishable from the surrounding tissues. Hence to reduce bleeding during surgical excision of the tumor the mass should be peeled off from its attachments and the mass should not be broken into.
Microscopic appearance: Microscopically the picture is of vascular spaces of varying shapes and sizes within a stroma of fibrous tissue. The relative proportions of the vascular and the stromal components change with the age of the swelling. In earlier lesions the vascular component stands out as an all pervasive feature, whereas in the more long standing tumors collagen predominates. It could also be seen that, as one strays away from the heart of the tumor the fibrous tissue element overshadows vascular element. The tumor is covered by squamous epithelium. In some cases pseudostratified columnar epithelium is seen side by side with the metaplastic squamous epithelium.
Cellular infiltration is a common feature, particularly in the superficial parts of the tumor, underneath the epithelium. the infiltrates include, plasma cells, lymphocytes, polymorphs and eosinophils in varying proportions.
The structure of the tumor is made up of fibrous tissue elements and vascular channels. The fibrous tissue element is made up of cells which are spindle shaped, oval or round cells. In between these cells, the bundles of collagen fibres are seen running in different directions.
The vascular channels of the tumor may be divided into 2 main types. One type which is seen in all cases is made up of spaces lined with one layer of endothelium and is free of any muscular coating. In the other type the vascular channels are made up of arteries with thick muscular coating.
Mucous glands may be seen in the superficial parts of the tumor underneath the epithelial covering. In some patients nerve bundles could also be seen. In long standing tumors, there is a tendency towards gradual compression of the sinusoids so that the lining endothelial cells are pushed against each other like cords, where in others intravascular thrombosis occur.
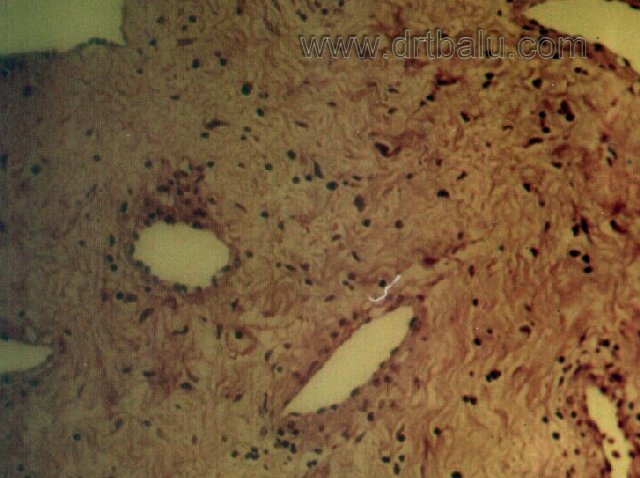
Photomicrograph showing histological features of JNA
Clincial features: Signs and symptoms are present for an average of 6 months prior to the diagnosis, commonly with extension beyond the nasopharynx.
The two cardinal symptoms of angiofibroma are nasal obstruction and intermittent unprovoked epistaxis. Epistaxis may vary in severity from an occasional show to an alarming sometimes threatening torrent. Chronic anaemia is thus a feature of an established JNA.
The nasal obstruction is so complete causing stasis of secretions and sepsis become inevitable. Patients may even have hyposmia or anosmia.
The voice of the patient acquires a nasal intonation. If the swelling enlarges to force the soft palate down, the voice may become plummy.
Blockage of eustachean tube orifice is also common causing deafness and otalgia. Headache is not uncommon in long standing cases. If present it could be attributable to chronic sinusitis in some patients. Intracranial extension of the mass could also be the cause for headache in these patients.
Diplopia may occur secondary to the erosion of the mass into the cranial cavity and causing pressure on the optic chiasma. Failing vision has been attributed by Shaheen to the tenting of the optic nerveby the tumor.
Anterior rhinoscopy shows the presence of abundant purulent nasal secretions together with bowing of nasal septum to the uninvolved side. Posterior rhinosocpy in a cooperative patient shows a pink or red mass filling the nasopharynx. Due to the bulk of the lesion it may not be always possible to ascertain the site of origin accurately.
Gross physical signs become evident when extensive disease involves the nose and infra temporal fossa. The nasal bones become spayed out and there may be obvious swelling in the temple and cheek. Intra oral palpation in the interval between the ascending ramus of the mandible and the side of the maxilla may reveal the tell tale thickening of disease which has crept around the back of the antrum. Impaction of bulky mass in the infra temporal fossa results in extreme signs, such as trismus and bulging of the parotid gland. Proptosis is a definite sign that the orbital fissures have been penetrated. The classic frog face seen in patients with extensive disease is due to massive escape of the disease.
Extension of tumor:
As growth enlarges it has to follow the lines of least resistance.
a. It hangs down in the cavity of nasopharynx and when large enough, it may depress the soft palate and may even peep below it.
b. It can work its way in to the corresponding nasal passage towards the anterior nares. It can cause pressure on the outer wall as well as on the septum bending it to the opposite side. The corresponding turbinates and ethmoidal air cells and the related antral wall may suffer pressure atrophy. The most common deformity referred to as the "frog face" is due to the forward spread involving the ethmoidal region. Lateral spread in to the maxillary sinus may be responsible for the cheek swelling.
c. The mass commonly arises from the sphenopalatine foramen area. It may have two components, one filling the nasopharynx and the other extending out into the pterygopalatine and infratemporal fosse. The central stalk joining the two components occupy the sphenopalatine foramen at the upper end of the vertical plate of palatine bone.
d. It can encroach into the orbit by passing through the infra orbital fissure.
e. It can erode the skull base and cause intracranial problems.
Blood supply of JNA: is from the
1. Enlarged maxillary artery
2. Ascending pharyngeal artery
3. Unnamed branches from internal carotid artery
Staging of the tumor: Staging of the tumor has been done to for prognosis and therapeutic approaches. The currently accepted staging has been devised by Andrews.
Andrews staging classification:
Stage I: Tumor limited to the nasal cavity
Stage II: Tumor extension into the pterygopalatine fossa, or maxillay, sphenoid or ethmoid sinuses.
Stage IIIa: Tumor extension into the orbit without intracranial involvement.
Stage IIIb: Stage IIIa with extradural (parasellar) intracranial involvement
Stage IVa: Intradural without cavernous sinus, pituitary, or optic chiasm involvement
Stage IVb:Involvement of the cavernous sinus, pituitary, or optic chiasm
Surgery is usually recommended for stages up to IVa while for stage IVb radiotherapy is advisable.
Investigations: Standard X-rays of paranasal sinuses may reveal opacity in the nose and sinus areas. Xray skull lateral view may show the mass inside the nasopharynx. CT scan has replaced the preferred imaging modality in these patients. Both plain and contrast CT scans must be performed to clinch the diagnosis. It also reveals the extent of the lesion and also helps in staging of the disease. MRI reveals the precise extent of the mass.
Carotid angiogram may be performed to identify the feeder vessel and also to embolise the feeder to reduce bleeding during surgery.
Differential diagnosis: JNA should be differentiated from:
1. Pyogenic granuloma
2.Choanal polyp
3. Angiomatous polyp
4. Nasopharyngeal cyst
5. Chordoma
6. Carcinoma
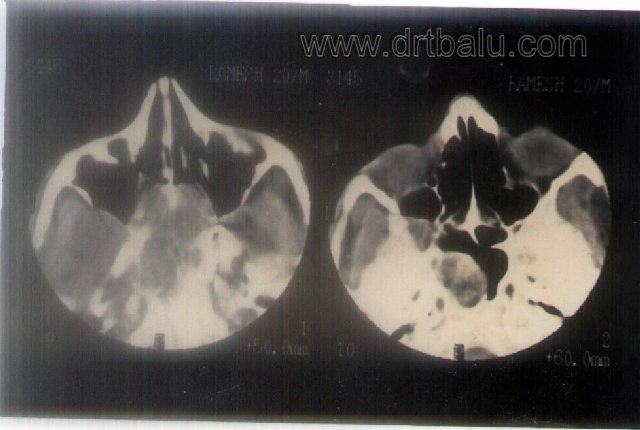
Axial contrast CT showing JNA mass in nasopharynx and sphenoid sinus
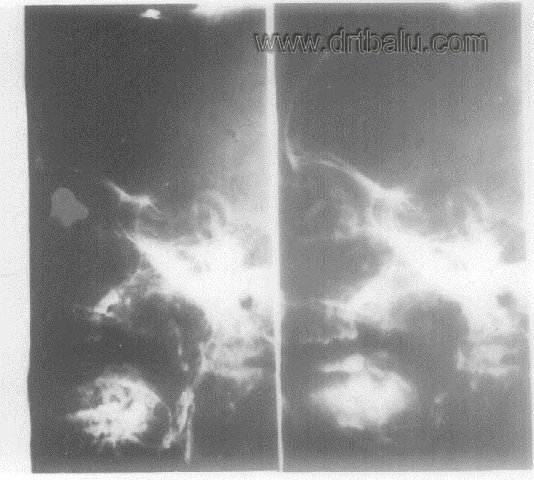
Carotid angiogram of a patient with JNA showing tumor blush
Treatment: Depends mainly on the extent of the lesion. Surgery is the preferred modality of treatment for all stages of the mass up to stage IVa while radiotherapy is used for stage IVb. Mainly three lines of treatment are available:
1. Surgery
2. Irradiation
3. Hormonal (purely supportive in nature)
Surgery: Complete excision of an extensive JNA mass is a desirable goal but is a surgical challenge because of the limited field of work, inadequate visualisation and profuse bleeding during surgery. Besides the deformity, scars and adhesions as a result of prior surgery adds to this problem. Currently several approaches are available to access the neoplasm. They are:
I Tumor removal - via naturalis:
This approach is preferred for very small tumors confined to nasopharynx. The tumor can be removed by subperiosteal dissection after soft palate retraction. Access is limited in this approach.
II Transpalatal approach:
Wilson in 1951 described this approach. This approach gives exposure to nasopharynx as well as extensions into the sphenoid sinus and choana. It gives no visible scar and post op healing is good. This approach is useful in dealing with masses in the nasopharynx with minimal extension into the choana and sphenoid sinus.
Procedure: Patient is put in tonsillectomy position. A forward curved incision is made just in front of the junction of hard and soft palate. Mucoperiosteum is separated either way. Posterior spine of the hard palate is removed. Incision is extended laterally and downwards on either side along the pterygomandibular raphe. The mucosa of the lateral pharyngeal wall is not divided and care is taken not to damage the greater palatine vessels. A good view of nasopharynx is achieved in this procedure. The mucous membrane on the side of the growth is incised with a blunt knife. Thus with blunt dissection the periosteum is elevated, growth is separated and finally avulsed in one piece.
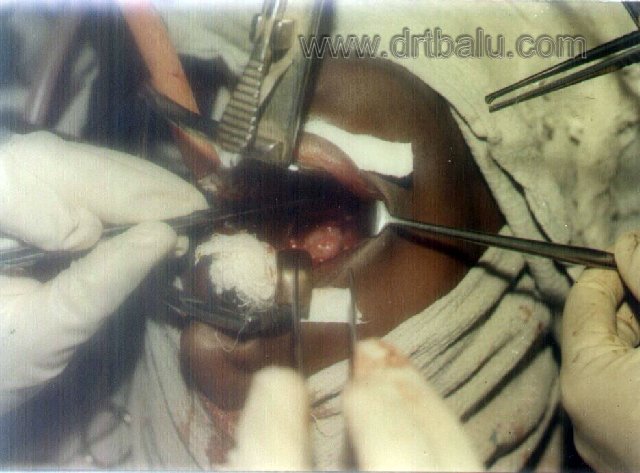
Image showing Wilson's transpalatine approach for JNA excision
III. Lateral rhinotomy approach: This approach is suited for smaller growth restricted to the nasal cavity. It is contraindicated for larger masses and whose extensions and attachements cannot be ascertained.
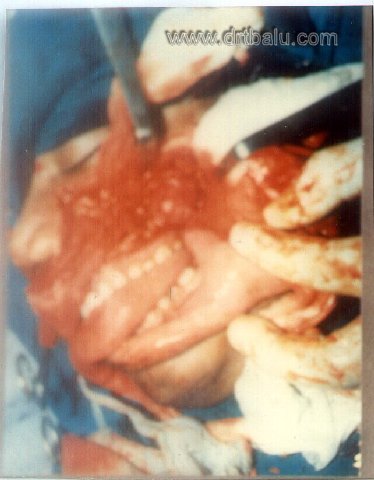
Image showing lateral rhinotomy approach
IV. Trans hyoid approach: This is suitable for tumors localised to nasopharynx without any extension into the surrounding structures. The major disadvantage is that it requires a temporary tracheostomy.
V. Transmandibular approach: (Kermen)
Incision is made vertically infront of the ear and carried down the neck anterior to the sternomastoid muscle. Dissection is started in the neck by exposing the carotid bifurcation at which level the external carotid artery is ligated. Lower pole of parotid is dissected free. The insertions of masseter muscle is severed from the mandible. Lateral aspect of the mandible is exposed by elevation of the periosteum. Transection of mandible is done with a giglisaw at a point 1 cm below the notch formed by coronoid and condyloid processes. Separation of mandible exposes the tube muscular wall of nasopharynx which is incised longitudinally on its lateral wall so that its lumen is entered. Tumor is exposed and dissected out.
VI. Sublabial midfacial degloving approach (Conley 1979): It is a bilateral extended trans nasal maxillary approach. There is no visible scarring, adequate exposure of nasal complex, nasopharynx and middle third of the face is obtained. Sublabial incision is performed from one maxillary tuberosity to the other. Intercartilagenous incisions are given to separate soft tissue of the nose from the upper lateral cartilages. Incisions along the pyriform aperture connects the circumferential septal vestibular incisions to the sublabial incision. This allows total mid facial degloving up to the roof of the nose and infra orbital foramen. The complication of this procedure is vestibular stenosis.
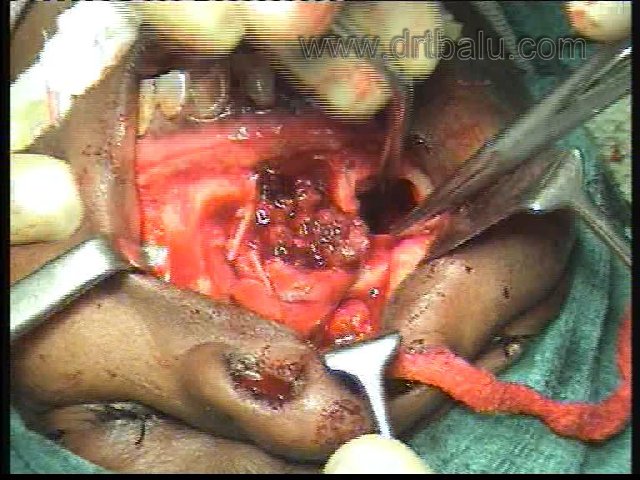
Image showing midfacial degloving approach
VII. Transzygomatic approach (Sami & Girgis 1965): This approach is useful for removal of tumor involving the temporal and infra temporal regions.
Combined approaches:
Depending on the size and extent of the mass a combination of various approaches can be attempted to extripate the mass in toto.
I. Transpalatal sublabial approach (Saldana 1965): This approach is useful for tumors extending into pterygoid and infratemproal fossa. A sublabial incision 'S' shaped is made extending to the maxillay tuberosity. After elevation of mucoperiosteal flap, the greater palatine neurovascular pedicle is preserved. The surgeon's index finger is inserted into the pterygopalatine fossa and blunt dissection is used to free the tumor from its lateral attachement. The mass can be deliverd via the nasopharynx.
II. Combined transpalatal and lateral rhinotomy approach: This approach is indicated for larger mass, recurrent JNA, and when attachement and extensions of the tumor are not predetermined. This approach gives excellent exposure and so the chances of recurrence are minimal.
III. Triple approach of Hiranandani (1968): In this approach transpalatal and lateral rhinotomy are combined along with caldwel luc. Complete exposure of pterygopalatine fossa is possible by removal of posterior wall of the maxillary antrum, after opening the antrum through caldwel luc incision. Chances of recurrence are minimised. Ligation of internal maxillary artery is done to reduce the bleeding.
Radiotherapy:
Radiotherapy can produce some amount of tumor regression by radiation vasculitis and occlusion of vessels by perivascular fibrosis. Radiotherapy should be reserved for selected patients such as those with inoperable intracranial extensions and recurrent tumors.
Disadvantages of radiotherapy:
a. If the child is exposed to large doses i.e. above 5000-6000 rads, there may be damage to eyes, spinal cord and brain.
b. Small doses are ineffective in reducing the blood supply or the size of the mass.
c. Radiotherapy may cause fibrosis and adhesions of surrounding tissue. Later surgery upon these patients becomes difficult.
d. Sarcomatous changes can occur in the mass as a result of irradiation.
Adjunctive treatment:
Hormonal therapy: Since JNA has been postulated as an endocrine tumor testosterone receptor blocking drugs / estrogens have been tried to reduce the mass. These hormones cause disaggreable side effects such as increased breast size. Hormones could even act as carcinogens.
Embolisation: Was first attempted by Robertson in 1972. This was not meant to be therapeutic measure. After embolisation bleeding is minimised during surgery. It is ideally carried out a few days before surgery. Hence it is a valuable preliminary to surgery. The feeding vessels to the tumor is identified. It is then deliberatly occluded by means of materials injected through a selectively placed catheter.
Materials used:
Autologous substances like fat, blood clot, or chopped muscle fragments.
Artifical materials: Gelfoam, Oxidised cellulose, Tantalum powder, glass beads, polyvinyl alcohol etc.
Embolisation should always be preceded by angiography. Subtraction films may be helpful in areas containing complex bony structures.
Immediate complications of embolisation are pain, embolisation of normal vessels, hypersensitivity. Delayed complications include fever, pain and infections.
Cryosurgery and Lasers can also be used during surgery to minimise bleeding.
